Explore advanced replicate study designs for bioequivalence assessment of highly variable drugs, including RSABE and full vs. partial design strategies.
Tag: bioequivalence standards
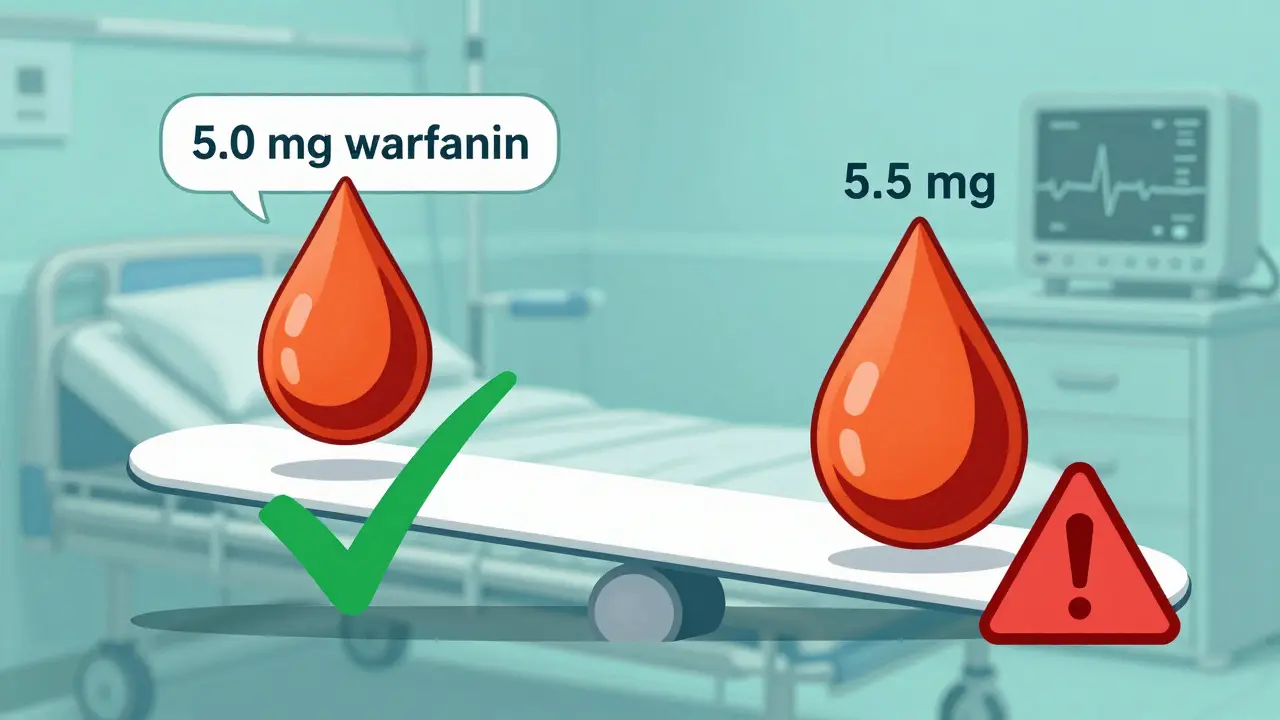
The FDA enforces stricter bioequivalence standards for NTI drugs like warfarin and phenytoin, requiring tighter limits (90-111%) and scaled testing to ensure safety. These rules prevent dangerous dosing errors in medications where small changes can be life-threatening.