When you pick up a generic medication at the pharmacy, you're trusting that it works exactly like the expensive brand-name version. But how do regulators actually prove two drugs are the same? For decades, the industry has leaned on pharmacokinetic studies is a method of measuring how a drug moves through the body, specifically its absorption, distribution, metabolism, and excretion. While often called the "gold standard," the reality is more nuanced. It's less of an absolute truth and more of a scientifically validated surrogate that tells us if a generic is "close enough" to be interchangeable.
The Blueprint for Generic Approval
Back in 1984, the U.S. FDA changed the game with the Hatch-Waxman Act. Before this, generic makers had to redo massive clinical trials to prove safety and efficacy-a process that was slow and incredibly expensive. The Act created an "abbreviated" pathway. Instead of proving the drug works from scratch, manufacturers only need to prove pharmaceutical equivalence -meaning the generic has the same active ingredient, strength, dosage form, and route of administration as the original.
Once that's settled, the heavy lifting moves to bioequivalence testing. For most pills you swallow, this means running a comparative study in humans to see if the generic drug reaches the bloodstream at the same rate and to the same extent as the innovator product. If the blood levels match up, the FDA assumes the clinical effect will also match.
The Numbers Game: Cmax and AUC
To prove equivalence, researchers don't just look at a graph; they hunt for two specific metrics. First is Cmax , which is the maximum plasma concentration-basically, the highest peak of the drug in your blood. Second is AUC (Area Under the Curve), which represents the total exposure of the drug over time.
The regulatory "magic number" is the 90% confidence interval. For a generic to pass, the ratio of the generic's Cmax and AUC compared to the brand name must fall between 80% and 125%. If it's within this window, it's generally considered therapeutically equivalent. However, for Narrow Therapeutic Index (NTI) drugs-medications where a tiny dose change can be toxic or ineffective, like warfarin or digoxin-the rules are much stricter. In these cases, the window might shrink to a tight 90-111% to ensure patient safety.
| Drug Type | Key Metrics | Standard Acceptable Range (90% CI) | Focus Area |
|---|---|---|---|
| Standard Systemic Drugs | Cmax & AUC | 80% - 125% | General Bioavailability |
| NTI Drugs (e.g., Warfarin) | Cmax & AUC | 90% - 111% | Patient Safety/Toxicity |
| Immediate-Release | Dissolution Profile | ≤10% difference | In vitro release rate |
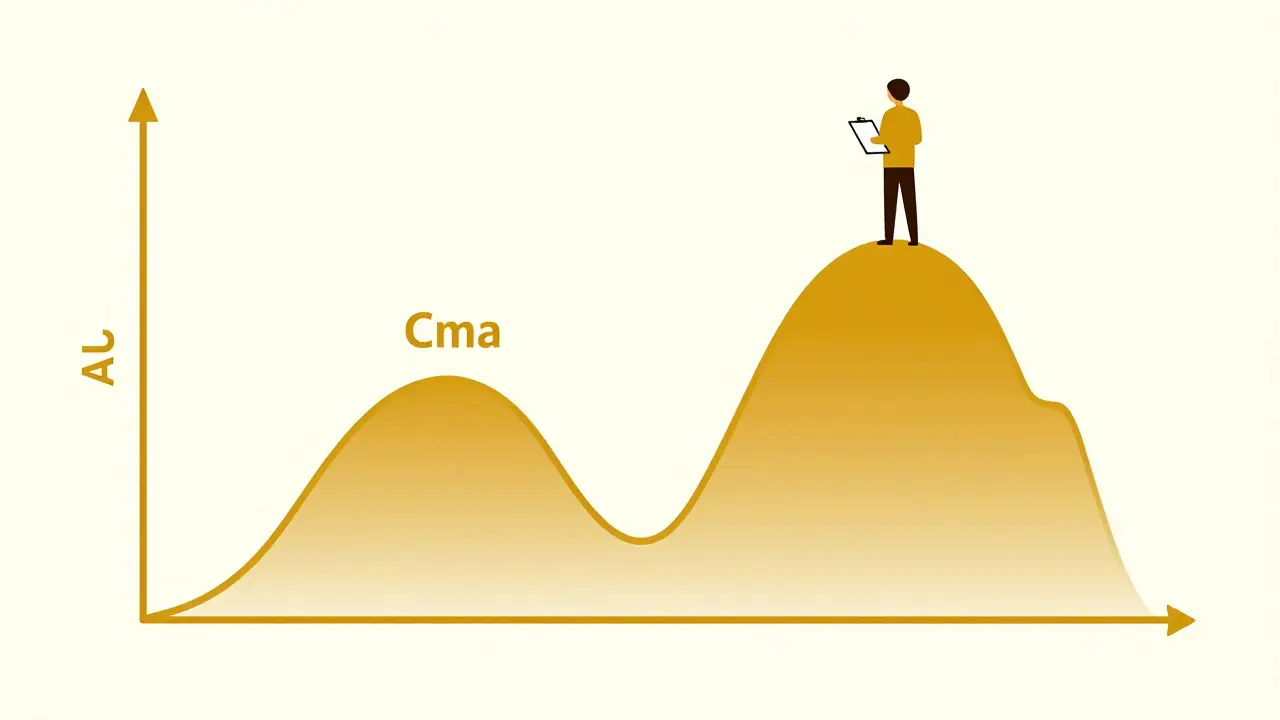
Where the "Gold Standard" Hits a Wall
If pharmacokinetic studies are so great, why is there a debate? Because for some drugs, blood levels don't tell the whole story. Take topical creams or patches. Measuring how much of a skin cream ends up in the blood is often useless because the drug is supposed to stay in the skin to work. In these cases, traditional blood tests are a poor proxy for efficacy.
There are even scarier examples. Research on gentamicin generics showed that some products were pharmaceutically equivalent and passed in vitro tests, yet they failed in vivo (in the actual body) regardless of who manufactured them. This proves that having the same ingredients doesn't always guarantee the same clinical outcome. This is why some experts argue that in vitro studies-tests done in a lab without humans-can actually be more reliable for certain immediate-release drugs because they remove the huge variability of human biology.

The Cost and Struggle of Proving Equivalence
For a pharmaceutical company, proving bioequivalence is often the most stressful part of development. It's not just a formality; it's a massive technical hurdle. A single bioequivalence study can cost anywhere from $300,000 to $1 million, and the process can take up to 18 months.
The difficulty spikes with modified-release formulations. If a company changes a single inactive ingredient (an excipient), it can completely change how the drug is released into the body, even if the active drug is identical. To avoid some of these hurdles, some companies use the Biopharmaceutics Classification System (BCS) . If a drug is highly soluble and highly permeable (BCS Class I), they might get a waiver for some human testing, though this only applies to about 15% of drugs.
The Future: Beyond Simple Blood Tests
We are moving away from a "one size fits all" approach. The FDA's Complex Generic Drug Products Initiative is creating specific guides for tricky drugs, acknowledging that a simple blood draw isn't always enough. One of the most exciting shifts is toward PBPK Modeling (Physiologically-based Pharmacokinetic modeling). Instead of just testing 24 people, scientists use computer models to predict how a drug will behave across different populations. Since 2020, the FDA has started accepting these models to support waivers for certain drugs.
For skin-related drugs, dermatopharmacokinetic methods are replacing old-school clinical trials. Instead of recruiting 500 patients to see if a rash clears up, researchers can use cryopreserved human skin to see exactly how the drug permeates. It's faster, more accurate, and avoids the unpredictability of human clinical endpoints.
Is a generic drug exactly the same as the brand name?
Not exactly. They must be pharmaceutically equivalent (same active ingredient, dose, and form), but they can have different inactive ingredients (excipients). Bioequivalence studies ensure that despite these differences, the drug behaves the same way in your body.
What happens if a drug falls outside the 80-125% range?
If the Cmax or AUC falls outside this range, the drug is not considered bioequivalent. The manufacturer must either reformulate the drug to change its absorption rate or conduct more extensive clinical trials to prove it still works safely.
Why are NTI drugs treated differently?
Narrow Therapeutic Index (NTI) drugs have a very small window between a dose that works and a dose that is toxic. Because the risk is higher, regulators use a tighter equivalence range (like 90-111%) to ensure there is almost no variation between the generic and the brand.
Can a drug be pharmaceutically equivalent but not therapeutically equivalent?
Yes. As seen with some gentamicin generics, you can have the exact same active ingredients and pass lab tests, but the drug may still fail to produce the same therapeutic effect in a living patient due to complex biological interactions.
What is the role of the World Health Organization (WHO) in this?
The WHO sets international standards for therapeutic equivalence. They define it as two products having essentially the same efficacy and safety when given the same dose, providing a framework that many national regulatory agencies follow to harmonize their rules.

william wang
April 15, 2026 at 21:06This is a great breakdown of a complex topic. It's really interesting how the 80-125% window is the baseline, because for most people, that seems like a huge gap, but in terms of plasma concentration, it's the accepted standard for safety and efficacy. I've always wondered about the NTI drugs, and the 90-111% range makes a lot of sense given how dangerous a small fluctuation can be with something like warfarin.